Galfan lasts 2-3 times longer than galv
Gafan coating of 275grams/m2 (ZGF 275 spec)
Galfan is 95% Zinc 5% Aluminium and lasts 2-3 times longer than Galv which is 100% Zinc
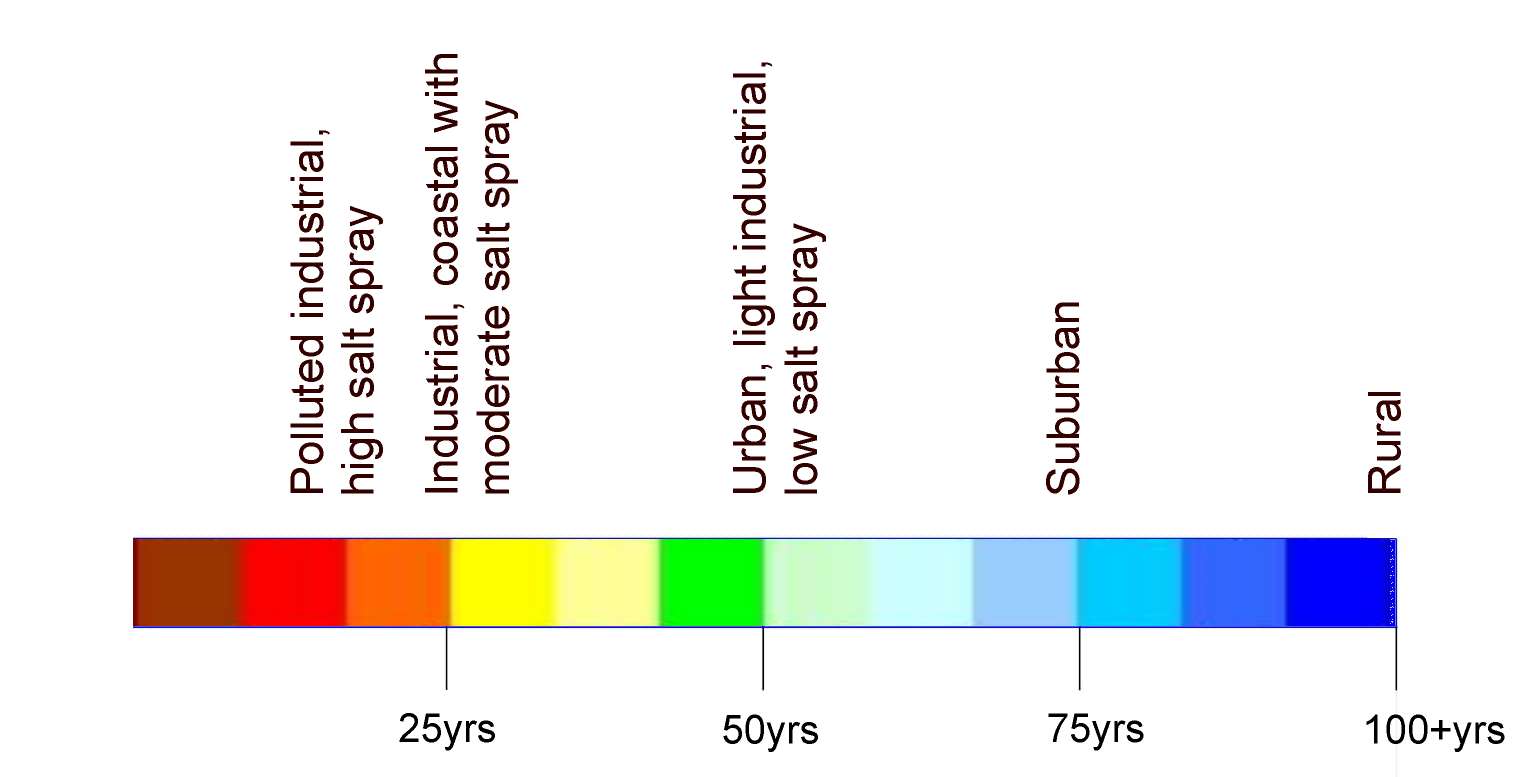
Expected Life Expectancy
Time to first maintenance or DBR, is defined as the time to 5% rusting of the steel surface.
The gabion structure at 5% rust still holds the rocks in place, so is still structurally sound, for many years after 5%DBR (Dark brown rust) stage.
Good drainage.
Good drainage behind and below your gabion retaining wall will not only improve the retaining walls stability, it will also increase the expected design life of the gabion wall
EN ISO 9223
Environment Harshness table
| Life Span | |
|---|---|
| C1: Very Low (Internal buildings with clean atmospheres) | 100 years + |
| C2: Low (Low level of pollution, mostly rural areas) | 100 years + |
| C3: Medium (Urban and industrial atmospheres, moderate sulphur dioxide pollution, coastal areas with with low salinity) | 50 years + |
| C4: High (Industrial areas and coastal areas with moderate salinity) | 30 years + |
| C5: Very High (Industrial areas with high humidity and aggressive atmosphere, coastal and offshore areas with high salinity within 500m of the sea) | 15 years + |
| CX: Extreme (Extreme industrial areas, coastal and offshore areas with prolonged contact with salt spray) | 5 years |
Special Environments.
In New Zealand there are areas where local conditions create an increased likelihood of corrosion. Special consideration should be given to material selection and design in these areas. They include:
1. Geothermal Areas
Hydrogen sulphide associated with geothermal activity creates a corrosive environment. Variations in natural activity or draw-off from steam bores plus the effects of weather conditions make the high risk areas hard to define.
2. West Coast, South Island
In this area, smoke from the coal burning fires may cause high concentrations of sulphur dioxide in the air. The combination of this and the high rainfall for the region creates an aggressive situation which must be considered . The effects of a severe coastal environment aggravate the situation. This area combines the most severe features of both industrial and coastal environments.
3. Coastal Environment
Within 1000m of the coast, characterised by:
Light salt deposits
A frequent smell of salt in the air
In the immediate vicinity of large expanses of calm salt water such as harbour foreshores
This environment may be extended inland by prevailing winds and local conditions.
4. Industrial Environments
Within 500m of corrosive industrial emissions and subject to heavy fallout from them.
Acidic Soils.
When constructing gabion retaining walls in strongly acidic soils (PH over 5.5), the soil and the corrosive groundwater must be separated from the gabion structure by using geotextile fabrics and a properly designed drainage system.
AS /NZS 4534.
Zinc and zinc/aluminium – alloy coatings on steel wire Class W10 (heavy coating).
15 year Study.
Here is the link to a 15 year long study on factors that effect the life span of gabions
Galvanisation.
Galvanising protects the gabion steel in two ways
1/ it forms a corrosion resistant coating over the steel, that protects the steel from oxidation.
2/ the galfan serves as a sacrificial anode, so even if the coating is scratched the exposed steel, does not rust while the ZnAl is present
Aluminium and zinc are used as sacrificial metals. They are more reactive than iron and lose their electrons in preference to iron. Therefore there is no need to protect the cut ends of the gabion wire, or worry about small scratches to the gabion during construction.
PVC Coating.
1/ PVC coating does not improve the gabions resistance to damage from flood debris impact.
Rock impact tends break wires instantly and the thicker the wire the less likely this damage is to occur.
Gabion1 recommend using the thicker 4.5mm welded mesh gabions for erosion control
Caltrans report states that,
State of California Gabion mesh corrosion report
PVC-coated wires usually performed better than zinc-coated wires in severe, natural exposures. They showed very little to no strength loss for about 13 years, although wires were in early states of corrosion. Because the bond between PVC and the underlying metal broke after cycling through temperature changes (daily or seasonally), a capillary space formed, where electrolyte migrated, and then corrosion started. In continued severe exposures, we expect that PVC-coated wires will continue to corrode, possibly disintegrating within the confined PVC sheath, or expanding and breaking the PVC as corrosion compounds form.
PVC has a smooth and shiny surface when it is newly manufactured. After about 3 to 5 years of exposure to sunlight, PVC photo-degraded, and it became dull and got chalky. Simply described, photons (incident light) displaced chloride ions in surface molecules. Plasticizers may be added to PVC for flexibility. After about 7 years at site 7, the surfaces of sun-exposed PVC coating developed cracks. As plasticizer compounds volatilized, PVC coating shrank and cracked. Among the sites where test panels were submerged in either mud or water, PVC did not become dull, like sun-exposed PVC. Instead, PVC hardened and often discolored by reacting with its surroundings.
Once the PVC coating is damaged, the gabion then relies on the Galfan coating to prevent corrosion.
In erosion control applications the PVC is often chipped off and damaged by flood debris
The images below are of PVC mesh that was damaged in transit, before gabion assembly and prior to the rocks being added.